 
This Flinn handout also gives important disposal instructions. In their experiment, the effect of temperature on the equilibrium shift is explored. 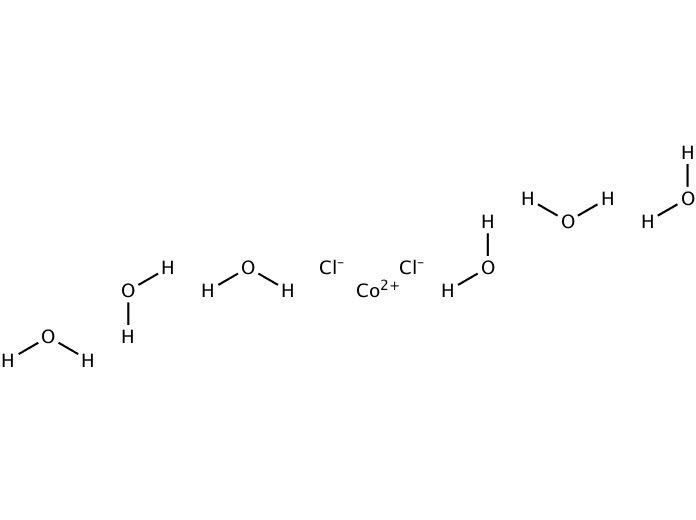
[Flinn has a PDF handout and video with a similar lab except acetone is not used. The following year, they concluded that it was worth the wait. After a discussion about the lab, the student decided to take grade 12 chemistry, just to experience it for themselves. In fact, once a grade 11 chemistry student asked about the experiment after eyeing the set of these test tubes in the fume hood where I was evaporating off the acetone. I really enjoy experiencing students' surprised reactions when the two colourful layers form. This lab comes from Chemistry: A Modern Course, Teacher Resource Book, Merrill (1987) - still enlightening students 30 years later. The difference in density is what allows the colour change to occur at the top of the test tube. The acetone undergoes hydrogen bonding with the H 2O, causing the equilibrium to shift to the right producing more of the blue CoCl 4 2- ion. The blue layer on top of a pink layer in the cover photo is accomplished by SLOWLY adding 10 mL of acetone toĥ mL of aqueous 0.2 M CoCl 2. A second sample of 0.416 g of the compound was dissolved in water. Students experiment with shifting the equilibrium and observing the colour change by The silver chloride was filtered, dried, and weighed, and it had a mass of 0.308 g. This classic Le Châtelier’s Principle lab explores the reversible chemical reaction:Ĭo(H 2O) 6 2+ + 4Cl – + heat -> CoCl 4 2– + 6H 2O The blue colour is the result of cobalt chloride complex ions (CoCl 4 2–) in less dense acetone. On the front cover, the pink colour in the test tube comes from cobalt(II) ions in water, Co(H 2O) 6 2+. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
